Immune cell phenotyping is an important capability for immunology and immuno-oncology research,...
Challenges in Clinical Sample Logistics
The Challenges in Clinical Sample Logistics
The rise of precision medicine has brought a surge in the complexity of clinical trial design. A recent analysis of clinical trials in oncology revealed that the share of clinical trials with biomarker-based endpoints grew from approximately 15% to 55% between 2000 and 2018.1 The same analysis showed a compound annual growth rate of more than 30% for clinical trials evaluating three or more biomarkers. Plus, with a growth in IO combination trials in oncology2—and the need for deep immune phenotyping required for these trials—there is no indication that this trend will slow down in the foreseeable future.
For those managing clinical programs, the paradigm shift in biomarker-based clinical trials presents a host of new challenges, including increasingly complex sample logistics. Many clinical sites do not have the infrastructure to perform the deep immunophenotyping and biomarker assays that most modern clinical trial designs require. So instead, PBMC samples are often collected and prepared at clinical sites, then cryopreserved and sent to a central lab where the specimens are thawed and analyzed. The trouble with this approach is that the process of cryopreservation introduces error. Even the most optimized protocols for freeze/thawing PBMCs can lead to inconsistent results, and in some cases, a complete loss of key immune populations stemming from damaged target epitopes.3
ChipCytometry
Many customers facing these challenges turn to Canopy Biosciences’ ChipCytometry, an end-to-end platform for quantifying high-plex protein biomarker assays in a variety of sample types. ChipCytometry enables the quantitative measure of virtually unlimited protein biomarkers with single-cell resolution in fluid samples like PBMCs, and tissue samples.

Figure 1. A. Photograph of a rack of four ZellSafe Chips. B. Cross section of the sample microfluidic channel in a ZellSafe Chip. Cells adhere to the surface of the channel via electrostatic interaction and immobilized through multiple rounds of staining with antibody cocktails.
Sample Collection with ZellSafeTM Chips
A pillar of the ChipCytometry platform is Canopy’s proprietary ZellSafeTM Chips, which are microfluidic-based sample biorepositories. Isolated PBMCs are simply transferred to the ZellSafe Chips, where they can be stored at +4 °C and transported to a central lab without degrading the quality of the sample, therefore eliminating the cryo-bias introduced by freeze/thaw protocols.
Long-term Sample Storage
In addition, Canopy Biosciences have validated biomarker stability for up to two years at +4 °C, when samples are stored on ZellSafe Chips. The extended sample stability enables repeated re-interrogation of samples. After analyzing a 15-plex assay, for example, the samples can be stored and interrogated for an additional set of markers at a later time—for up to two years.
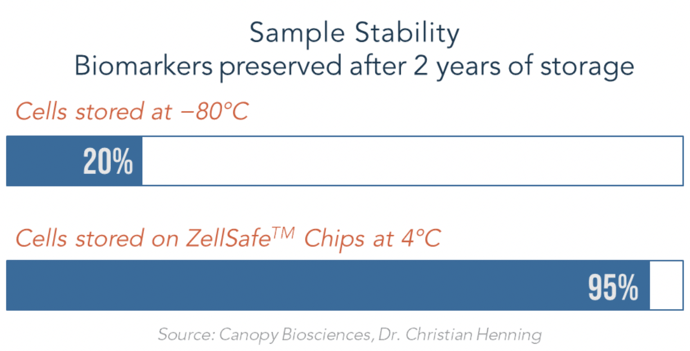
Figure 2. Samples stored on ZellSafe Chips are stable for up to 2 years. Meanwhile, cryopreserved samples undergo significant degradation of biomarkers over the same time period. ChipCytometry enables re-interrogation of samples to add additional markers to an assay after the initial analysis. This feature eliminates cryo-bias and is ideal for precious clinical samples.
To learn more about the revolutionary ZellSafe Chips, check out our 15-min webinar on clinical biobanking for ChipCytometry or contact us for more information.
References:
- Vedas, et al. L.E.K. Consulting, www.lek.com
- CancerMPact® Cancer Landscape, Life Sciences by Kantar, kantar.com
- Kadić et al. BMC Immunology (2017) 18:6